
Trump trial sees new witnesses to close out first week of testimony
Prosecutors in former President Donald Trump's criminal trial in New York called two new witnesses to the stand on Friday, rounding out the first week of testimony.
Watch CBS News

Prosecutors in former President Donald Trump's criminal trial in New York called two new witnesses to the stand on Friday, rounding out the first week of testimony.

With a relatively low average monthly cost of living and a low crime rate, this little-known town has a lot to offer retirees according to one report.

About 7 in 10 retirees stop working before they turned 65. For many of them, it was for reasons beyond their control.

A U.S. MQ-9 Reaper has crashed off Yemen's coast. It may be the third $30 million drone shot down by the Houthis since November.

An emergency exit slide "separated" from a Delta flight Friday, prompting an emergency return to New York City's John F. Kennedy Airport.

The discovery of drug-resistant bacteria in two dogs prompted a probe by the CDC and New Jersey health authorities.

A gold pocket watch recovered along with the body of John Jacob Astor, the richest passenger on the Titanic, is up for auction.

The White House had been due to decide on the menthol cigarette rule in March.

Border officers have broad authority to search travelers' electronic devices without a warrant or suspicion of a crime.

Harvey Weinstein is still serving 16 years in a California case, which is expecting an appeal that could use similar arguments to that of the overturned New York conviction.

Secretary of State Antony Blinken has been weighing whether to recommend suspending U.S. aid to an IDF unit under a measure known as the Leahy Law.
First known HIV cases from a nonsterile injection for cosmetic reasons highlights the risk of unlicensed providers.

Angel Gabriel Cuz-Choc was found hiding in a wooded area after his girlfriend and her 4-year-old daughter were found dead in Florida.

After Kristen Trickle died at her home in Kansas, her husband Colby Trickle received over $120,000 in life insurance benefits and spent nearly $2,000 on a sex doll supposedly to help him sleep.

There has been no confirmed evidence of a grizzly within the North Cascades Ecosystem in the U.S. since 1996.

A pair of bears picked the wrong person to mess with when they approached a 50-year-old karate practitioner.

The king took a break from public appearances nearly three months ago after he was diagnosed with an undisclosed type of cancer while he was undergoing treatment for an enlarged prostate.

Fans vote for the award winners - often leading to surprise winners and collaborative performances.

The court is considering whether Trump is entitled to broad immunity from criminal charges in the 2020 election case.

There are no cameras allowed in the court where he's being tried.

A federal judge rebuffed his request for a new trial in the civil suit that ended with an $83.3 million judgment for her.

The FBI calls on tech companies to "step up" to protect people looking for love online.

Laura Kowal's match on an online dating site wasn't what he seemed. Now her daughter wants to expose the risk of such scams.

Scammers have been increasingly successful in leveraging their romantic grip on victims.

"I am happy to debate him," President Biden said during an interview with Howard Stern.

Trump has in the past railed against absentee voting, declaring that "once you have mail-in ballots, you have crooked elections."

President Biden finds familiar and active allies for his reelection bid with labor union endorsements.
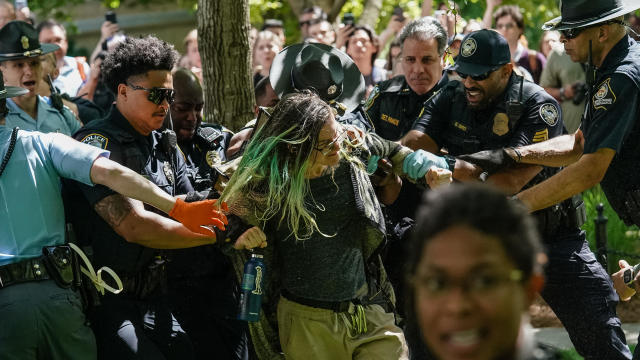
Police are cracking down at some university protests over Israel's war against Hamas in Gaza.
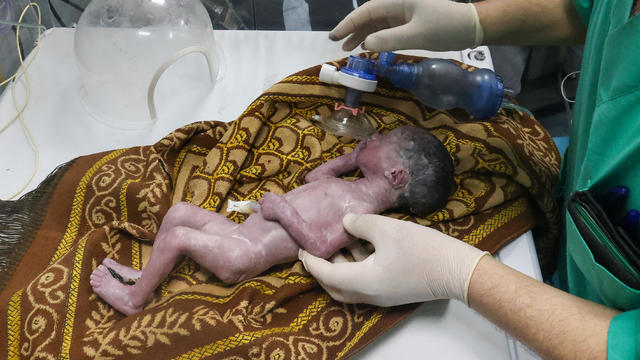
Sabreen Erooh had survived an emergency cesarean section after her mother was fatally wounded in an Israeli airstrike.

Senate Minority Leader Mitch McConnell appears on "Face the Nation" as pro-Palestinian protests roil American politics.

Gold can be a smart bet for seniors — and that's not just due to the recent gold price uptick, either.

A HELOC can be a great borrowing option now, but the repayment process is unique. Here's what to know about it.

The average American is currently facing a hefty amount of credit card debt — but there are good ways to tackle it.

With a relatively low average monthly cost of living and a low crime rate, this little-known town has a lot to offer retirees according to one report.

About 7 in 10 retirees stop working before they turned 65. For many of them, it was for reasons beyond their control.

Sabrina Bishop, a live-in caregiver for an older man with advanced dementia, is just one of the many people in her profession facing financial challenges.

A gold pocket watch recovered along with the body of John Jacob Astor, the richest passenger on the Titanic, is up for auction.

The median mortgage payment jumped to a record $2,843 in April, up nearly 13% from a year ago, a new analysis finds.

Got bees? Don't put up with all that buzzing and stinging; here are some easy ways to get rid of bees this spring.

Save big on appliances from top brands and retailers gearing up for Memorial Day.
Discover the best deals on all kinds of Apple iPad models, allowing you to snag a new tablet and save some cash.

Former National Enquirer publisher David Pecker's testimony will continue in Donald Trump's New York criminal trial Friday morning, a day after he revealed new details about the alleged "catch and kill" scheme he engaged in for the Trump campaign in 2016. CBS News correspondent Errol Barnett has more.

Secretary of State Antony Blinken met with Chinese President Xi Jinping Friday in an effort to stabilize relations between the U.S. and China. While speaking in Beijing earlier, Blinken urged China to end its support for Russia's war in Ukraine. CBS News national security reporter Olivia Gazis has more.

An American tourist in Turks and Caicos is out on bail after he was arrested by airport security when they allegedly found ammo in his luggage. Ryan Watson says it was mistakenly in his bag, but he's now facing a potential mandatory minimum sentence of 12 years behind bars. CBS News senior travel adviser Peter Greenberg has more.

Reality star and designer Whitney Port discusses her new partnership with prenatal vitamin company Perelel and launches the "Fertility, Unfiltered" video series. She also talks for the first time about her personal decision to pursue IVF again after facing challenges in conceiving a second child.

Democratic Congressman Ro Khanna joins The Takeout to discuss President Biden's approach to immigration, the economy & conflict in the Middle East. Khanna talks Biden's outlook in the 2024 election, America's electoral future & a teacher who inspired him.

Author and former Golf Channel broadcaster Lisa Cornwell talks to Major Garrett about her book, "Troublemaker," detailing her experience at the network. They discuss the state of the PGA compared to the LPGA.

CBS correspondent and author Jonathan Vigliotti joins The Takeout to discuss his new book "Before It's Gone," about how climate change is impacting small towns due to their lack of infrastructure and financial resources. He focuses on how communities rebuild after climate-related disasters and what lessons can be learned from their resilience.

For this edition of "The Takeout," Major Garrett speaks to filmmaker Brian Knappenberger about directing the nine-part documentary series, "Turning Point: The Bomb and the Cold War." The Netflix series offers a comprehensive look at the Cold War and its aftermath. Knappenberger says the aim of his documentary was to reflect and to also explore present-day tensions.

Analyst and author Ken Block joins The Takeout to discuss his new book, "Disproven." Block explains his hiring by the Trump campaign to search for voter fraud, his fact-driven investigation into claims of voter fraud and where the claims may have come from.

Sophia Bush filed for divorce from entrepreneur Grant Hughes in August 2023 after a year of marriage and started dating the former world champion soccer player afterward.

The Heisman Trophy was returned to former University of Southern California running back Reggie Bush Thursday after a 14-year dispute with the NCAA.

Reality star and designer Whitney Port discusses her new partnership with prenatal vitamin company Perelel and launches the "Fertility, Unfiltered" video series. She also talks for the first time about her personal decision to pursue IVF again after facing challenges in conceiving a second child.

Reggie Bush reflects on the reinstatement of his Heisman Trophy after 14 years, discusses his ongoing defamation lawsuit against the NCAA and shares his insights on the future of college football. This marks his first in-depth interview since the Heisman Trust's decision to return the award.

Police bodycam video shows the police encounter that ended in the death of Frank Tyson, a Black man in Canton, Ohio. The officers arrested him after a car crash and restrained him facedown. Warning, the video is disturbing.

In California, we dine out at a restaurant powered by robots. Then in Washington, we take a sip of a beanless cup of coffee, which aims to reduce the environmental impact of the popular beverage. Watch these stories and more on "Eye on America" with host Michelle Miller.

In New York, we tour a unique museum that’s home to an extensive collection of toys, games and playgrounds. Then, we sit down with NBA superstar Steph Curry to discuss his heartwarming new children’s book. Watch these stories and more on "Eye on America" with host Michelle Miller.

In Arizona, we learn why flag football is becoming an increasingly popular sport, especially among girls. Then in New York, we meet with descendants of some of the most notable suffragists of the 20th century. Watch these stories and more on "Eye on America" with host Michelle Miller.

In Connecticut, we meet the preservationists who are giving dilapidated lighthouses new life. Then in California, we learn about the efforts to restore an iconic fishing boat. Watch these stories and more on "Eye on America" with host Michelle Miller.

In Louisiana, we learn how a devastating drought has greatly diminished the area’s crawfish supply. Then in Ohio, we tour a small business that’s seeing promising results from a four-day work week model. Watch these stories and more on Eye on America with host Michelle Miller.

In 1961, Ed Dwight was selected by President John F. Kennedy to enter an Air Force training program known as the path to NASA's Astronaut Corps. But he ultimately never made it to space.

At his lowest moment, U.S. Army veteran and former teacher Billy Keenan found strength in his faith as he was reminded of his own resilience.

A surfing accident left New York teacher Billy Keenan paralyzed, but when he received a call from a police officer, his life changed.

The So Much To Give Inclusive Cafe in Cedars, Pennsylvania employs 63 people — 80% have a disability.

A mom was worried about what her son, who has autism, would do after high school. So she opened the So Much To Give cafe, a restaurant in Cedars, Pennsylvania, that employs people with disabilities – and helps them grow.

CBS Reports goes to Illinois, which has one of the highest rates of institutionalization in the country, to understand the challenges families face keeping their developmentally disabled loved ones at home.

As more states legalize gambling, online sportsbooks have spent billions courting the next generation of bettors. And now, as mobile apps offer 24/7 access to placing wagers, addiction groups say more young people are seeking help than ever before. CBS Reports explores what experts say is a hidden epidemic lurking behind a sports betting bonanza that's leaving a trail of broken lives.

In February 2023, a quiet community in Ohio was blindsided by disaster when a train derailed and authorities decided to unleash a plume of toxic smoke in an attempt to avoid an explosion. Days later, residents and the media thought the story was over, but in fact it was just beginning. What unfolded in East Palestine is a cautionary tale for every town and city in America.

In the aftermath of the Supreme Court striking down affirmative action in college admissions, CBS Reports examines the fog of uncertainty for students and administrators who say the decision threatens to unravel decades of progress.

CBS Reports examines the legacy of the U.S. government's terrorist watchlist, 20 years after its inception. In the years since 9/11, the database has grown exponentially to target an estimated 2 million people, while those who believe they were wrongfully added are struggling to clear their names.

Fans vote for the award winners - often leading to surprise winners and collaborative performances.

About 7 in 10 retirees stop working before they turned 65. For many of them, it was for reasons beyond their control.

Prosecutors in former President Donald Trump's criminal trial in New York called two new witnesses to the stand on Friday, rounding out the first week of testimony.

With a relatively low average monthly cost of living and a low crime rate, this little-known town has a lot to offer retirees according to one report.

Border officers have broad authority to search travelers' electronic devices without a warrant or suspicion of a crime.

About 7 in 10 retirees stop working before they turned 65. For many of them, it was for reasons beyond their control.

With a relatively low average monthly cost of living and a low crime rate, this little-known town has a lot to offer retirees according to one report.

The China-based owner of TikTok is facing a new law that will force it to either sell the wildly popular video platform, or face a U.S. ban.

First known HIV cases from a nonsterile injection for cosmetic reasons highlights the risk of unlicensed providers.

A gold pocket watch recovered along with the body of John Jacob Astor, the richest passenger on the Titanic, is up for auction.

Prosecutors in former President Donald Trump's criminal trial in New York called two new witnesses to the stand on Friday, rounding out the first week of testimony.

Border officers have broad authority to search travelers' electronic devices without a warrant or suspicion of a crime.

The White House had been due to decide on the menthol cigarette rule in March.

A U.S. MQ-9 Reaper has crashed off Yemen's coast. It may be the third $30 million drone shot down by the Houthis since November.

"I am happy to debate him," President Biden said during an interview with Howard Stern.

The White House had been due to decide on the menthol cigarette rule in March.

The discovery of drug-resistant bacteria in two dogs prompted a probe by the CDC and New Jersey health authorities.

First known HIV cases from a nonsterile injection for cosmetic reasons highlights the risk of unlicensed providers.

Are you using your smartwatch to the fullest? Here are 4 metrics doctors say can be useful to track beyond your daily step count.

Joel Embiid has been experiencing Bell's palsy symptoms, he said after Philadelphia's 125-114 win over the New York Knicks.

A U.S. MQ-9 Reaper has crashed off Yemen's coast. It may be the third $30 million drone shot down by the Houthis since November.
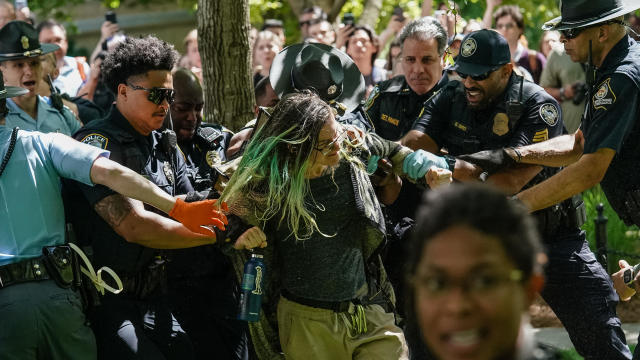
Police are cracking down at some university protests over Israel's war against Hamas in Gaza.

The king took a break from public appearances nearly three months ago after he was diagnosed with an undisclosed type of cancer while he was undergoing treatment for an enlarged prostate.

A gold pocket watch recovered along with the body of John Jacob Astor, the richest passenger on the Titanic, is up for auction.

Secretary of State Antony Blinken has been weighing whether to recommend suspending U.S. aid to an IDF unit under a measure known as the Leahy Law.

Fans vote for the award winners - often leading to surprise winners and collaborative performances.

Sophia Bush filed for divorce from entrepreneur Grant Hughes in August 2023 after a year of marriage and started dating the former world champion soccer player afterward.

Preview: In an interview to be broadcast on "CBS News Sunday Morning" April 28, the Oscar-nominated actress also talks about her debut as a singer-songwriter with the album "Glorious."

Looking for a place to live in NYC? Zillow is now listing Frank Sinatra and Mia Farrow's former home on the Upper East Side.

Italy's Culture Ministry has banned loans of works to the Minneapolis Institute of Art, following a dispute with the U.S. museum over an ancient marble statue believed to have been looted from Italy almost a half-century ago.

The Federal Communications Commission voted to adopt net neutrality regulations, a reversal from the policy adopted during former President Donald Trump's administration. Christopher Sprigman, a professor at the New York University School of Law, joins CBS News with more on the vote.

Are you using your smartwatch to the fullest? Here are 4 metrics doctors say can be useful to track beyond your daily step count.

From labor shortages to environmental impacts, farmers are looking to AI to help revolutionize the agriculture industry. One California startup, Farm-ng, is tapping into the power of AI and robotics to perform a wide range of tasks, including seeding, weeding and harvesting.

Local and federal authorities face challenges in investigating and prosecuting romance scammers because the scammers are often based overseas. Jim Axelrod explains.

U.S. regulators are reviving a rescinded rule, laying the groundwork for for a major court fight with the broadband industry.

Pediatrician Dr. Mona Hanna-Attisha, whose work has spurred official action on the Flint water crisis, told CBS News that it's stunning that "we continue to use the bodies of our kids as detectors of environmental contamination." She discusses ways to support victims of the water crisis, the ongoing work of replacing the city's pipes and more in this extended interview.

Ten years ago, a water crisis began when Flint, Michigan, switched to the Flint River for its municipal water supply. The more corrosive water was not treated properly, allowing lead from pipes to leach into many homes. CBS News correspondent Ash-har Quraishi spoke with residents about what the past decade has been like.

According to the University of California, Davis, residential energy use is responsible for 20% of total greenhouse gas emissions in the U.S. However, one company is helping residential buildings reduce their impact and putting carbon to use. CBS News' Bradley Blackburn shows how the process works.

Emerging cicadas are so loud in one South Carolina county that residents are calling the sheriff's office asking why they can hear a "noise in the air that sounds like a siren, or a whine, or a roar." CBS News' John Dickerson has details.

Representatives from across the world are gathering in Ottawa, Canada, to negotiate a potential treaty to limit plastic pollution. CBS News national environmental correspondent David Schechter has the latest on the talks.

Angel Gabriel Cuz-Choc was found hiding in a wooded area after his girlfriend and her 4-year-old daughter were found dead in Florida.

Dramatic bodycam footage shows the moment Florida deputies and K-9 dogs close in on a double murder suspect hiding in a thickly wooded area.

A new "48 Hours" investigation is looking into the death of a Kansas woman after she was found dying from a gunshot wound in 2019. The coroner initially ruled Kristen Trickle's death a suicide, but the local prosecutor said evidence on the scene didn't add up. "48 Hours" correspondent Erin Moriarty has the story.

A Bucharest court has ruled that a case against social media influencer Andrew Tate meets the required legal criteria and can go ahead, but there's no date set yet.

After Kristen Trickle died at her home in Kansas, her husband Colby Trickle received over $120,000 in life insurance benefits and spent nearly $2,000 on a sex doll supposedly to help him sleep.

Astronauts Barry Wilmore and Sunita Williams say they have complete confidence in the Starliner despite questions about Boeing's safety culture.

In 1961, Ed Dwight was selected by President John F. Kennedy to enter an Air Force training program known as the path to NASA's Astronaut Corps. But he ultimately never made it to space.
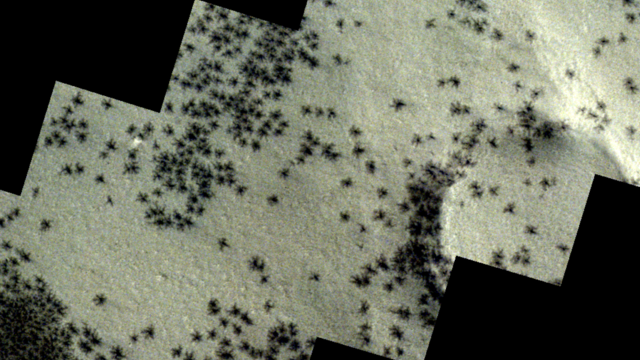
The creepy patterns were observed by the European Space Agency's ExoMars Trace Gas Orbiter.

The Shenzhou 18 crew will replace three taikonauts aboard the Chinese space station who are wrapping up a six-month stay.

In November 2023, NASA's Voyager 1 spacecraft stopped sending "readable science and engineering data."

A look back at the esteemed personalities who've left us this year, who'd touched us with their innovation, creativity and humanity.

The Francis Scott Key Bridge in Baltimore collapsed early Tuesday, March 26 after a column was struck by a container ship that reportedly lost power, sending vehicles and people into the Patapsco River.

When Tiffiney Crawford was found dead inside her van, authorities believed she might have taken her own life. But could she shoot herself twice in the head with her non-dominant hand?

We look back at the life and career of the longtime host of "Sunday Morning," and "one of the most enduring and most endearing" people in broadcasting.

Cayley Mandadi's mother and stepfather go to extreme lengths to prove her death was no accident.

Protests against the war in Gaza that have spread across U.S. college campuses are applying increased pressure on President Biden's administration over its continued support for Israel. CBS News chief White House correspondent Nancy Cordes has more on Mr. Biden's response to the demonstrations and an interview he did with talk-radio host Howard Stern.

Scott Pelley reports on America’s children of war, often overlooked, who live with disabled military veterans. Millions of kids are stepping up to help wounded warriors and their families deal with the emotional and physical trauma of service. Sunday.

Rhona Graff, Donald Trump's former executive assistant, took the stand Friday at the former president's New York criminal trial. CBS News' Errol Barnett has the latest on the trial.

The Federal Communications Commission voted to adopt net neutrality regulations, a reversal from the policy adopted during former President Donald Trump's administration. Christopher Sprigman, a professor at the New York University School of Law, joins CBS News with more on the vote.

President Biden met with Abigail Mor Edan, the youngest American hostage from the Oct. 7 attacks, at the White House Tuesday. Abigail's great-aunt, Liz Hirsh Naftali, joins CBS News with more details on Abigail's experience after being held in captivity.